New analytical methods reveal that fetuses encounter a far broader array of toxic “forever chemicals” in the womb than earlier tests suggested, raising fresh questions about exposure, monitoring, and health effects during development.
Scientists used more sensitive, wide-ranging chemical screening to look beyond the handful of well-known contaminants and found many more compounds crossing the placental barrier. The result is a snapshot of fetal exposure that is more complex and worrying than prior studies indicated. That complexity forces researchers to rethink how prenatal chemical burdens are measured and interpreted.
These substances belong to a class commonly called “forever chemicals” because they resist breakdown in the environment and in living tissue. They include per- and polyfluoroalkyl substances, or PFAS, which have been linked in past research to a range of health issues. Their persistence means once they enter bodies or ecosystems, they can hang around for years, and standard screens can miss less familiar variants.
The study applied advanced screening methods that cast a much wider net than routine targeted tests. Instead of checking for a short list of known PFAS, researchers used suspect screening and non-targeted analysis to detect unexpected or novel compounds. That approach revealed many molecular relatives and degradation products that earlier methods simply did not capture.
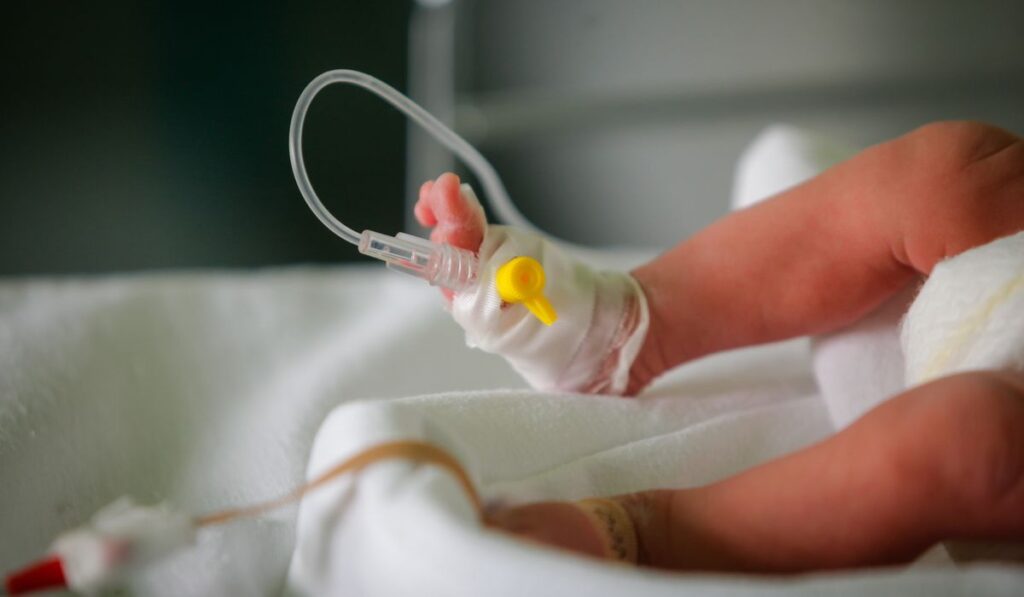
Sampling of placental tissue and cord blood was central to the investigation, showing direct fetal exposure rather than indirect inference from maternal samples alone. Detecting compounds in cord blood confirms passage across the placenta at a stage when the fetus is forming critical systems. Finding a broader suite of chemicals there raises immediate questions about timing, dose, and susceptibility during development.
Health scientists have long worried that early-life chemical exposures can alter immune response, hormone signaling, and organ development, and PFAS are no exception. Laboratory and epidemiological studies have linked some PFAS to changes in birth weight, immune function, and metabolic outcomes later in life. The discovery of more compounds in utero complicates efforts to pin down which specific chemicals or mixtures drive those effects.
Everyday sources help explain why so many different PFAS variants show up in human samples: contaminated drinking water, food packaging, stain- and water-resistant textiles, and industrial emissions all contribute. Consumers and workers can be exposed through ingestion, inhalation, and skin contact, and environmental pathways ensure pollutants move from products into air, soil, and water. That ubiquity means the fetus can be exposed even when pregnant people take care to reduce obvious risks.
The regulatory and monitoring landscape has struggled to keep pace with the diversity of PFAS chemistries. Many rules target a handful of long-studied compounds, leaving hundreds of related molecules unregulated and under-monitored. As analytical tools reveal more of these variants, policymakers and laboratories face a tougher job determining which chemicals deserve control and how to prioritize testing resources.
For researchers, the new findings change the questions and the methods they use. Instead of assuming a limited list of culprits, scientists must consider complex mixtures and unknown metabolites when designing toxicology studies and epidemiologic analyses. That shift means expanding chemical libraries, improving reference standards, and integrating exposure science with birth outcome research to better understand real-world risks.
Clinicians and public health professionals will also need clearer guidance on interpreting broader exposure profiles in pregnant patients and newborns. Right now, detecting additional PFAS variants raises concern but does not translate neatly into clinical action, given the uncertainty about which compounds pose the greatest harm. As the science advances, translating complex screening results into practical advice will be an essential but difficult step for protecting maternal and child health.