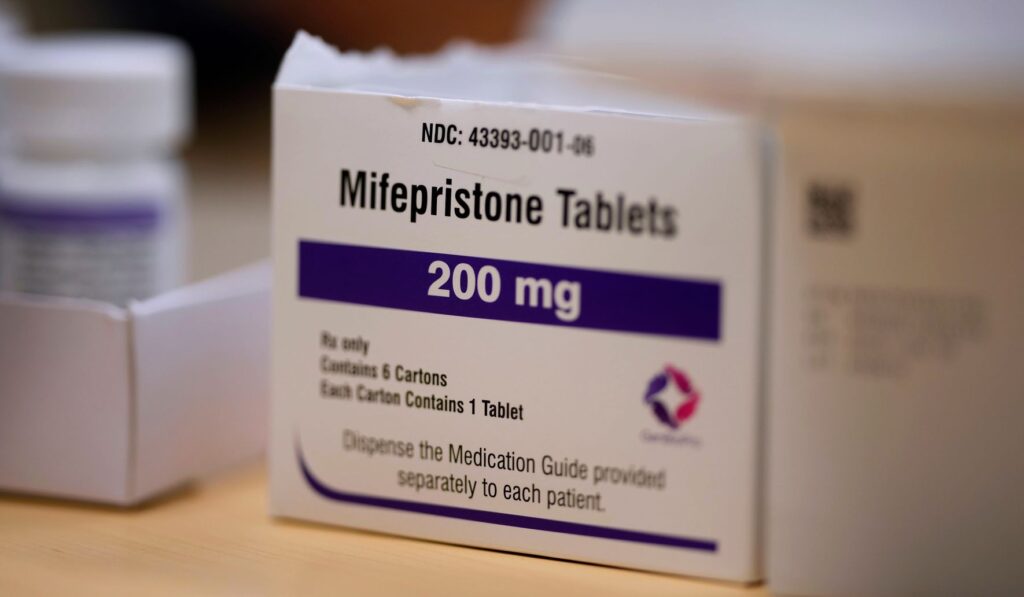
A federal appeals court has paused a policy that let mifepristone be prescribed through telemedicine and sent by mail, altering how the drug can be accessed nationwide and triggering immediate legal and practical fallout.
The court action halts a Biden-era decision that had allowed doctors to prescribe mifepristone remotely and pharmacies to ship it, rolling back a convenience that expanded virtual care. Supporters of the pause say it restores proper safeguards around a powerful medication, while opponents warn it disrupts medical care and burdens patients. The ruling moves control of access from a policy choice back into the courts and state systems almost instantly.
On its face this is about one medication and how it is distributed, but the implications are wider. Telemedicine had moved routine prescribing into living rooms and phones, and the court’s order interrupts that model for mifepristone specifically. Clinics, telehealth providers, and mail-order pharmacies now face uncertainty about whether they can continue long-established practices and how they should advise patients.
From a Republican perspective this decision underscores the need for clear, consistent regulation and enforcement when it comes to serious drugs. The push to expand remote access without what critics call sufficient safeguards raised legitimate concerns about safety, oversight, and adherence to original approvals. Restoring stricter controls is framed by supporters as enforcing the rule of law and preventing ad hoc policy changes from becoming permanent without review.
Providers and patients will feel the effects quickly in states with tighter restrictions. Women who relied on remote appointments and mail delivery may find themselves needing in-person visits, added travel, or new logistical hurdles to get a medication they previously obtained without leaving home. That shift will fall hardest on those with limited transportation, jobs with tight schedules, or in rural areas where clinics are sparse.
The legal battle is far from over and will play out in appeals, where judges will weigh administrative authority, public-health considerations, and statutory limits on agencies. Courts will look at how federal regulators approved or updated the policy and whether they followed proper procedure. For Republican policymakers, emphasis will be on judicial review and respecting the separation of powers when agencies change long-standing practices.
Meanwhile, pharmacies and telehealth companies must decide how to respond while avoiding liability. Some may pause remote prescribing for mifepristone to reduce legal risk, while others will try to maintain services under different protocols. That operational uncertainty forces quick compliance decisions and could push some businesses to limit offerings preemptively rather than face lawsuits or enforcement actions.
There are also public-health questions that courts and policymakers cannot ignore. Regulators must balance safety concerns about distribution against the potential harms of reduced access, including delayed care or unsafe alternatives. Republican arguments focus on ensuring that safety and oversight are not sacrificed for convenience, and on requiring that agencies act transparently when changing access rules for significant medications.
What happens next will depend on appeals and possible Supreme Court involvement, but the immediate practical picture is clear: a pause on remote prescribing and mailing of mifepristone creates disruption and forces new legal fights. Lawmakers on the right will press for clarity and oversight, while opponents will push back on access restrictions that they say harm patients. The case will shape how telemedicine is used for sensitive prescriptions going forward and will test how far administrative agencies can steer national health policy without fresh legislative direction.