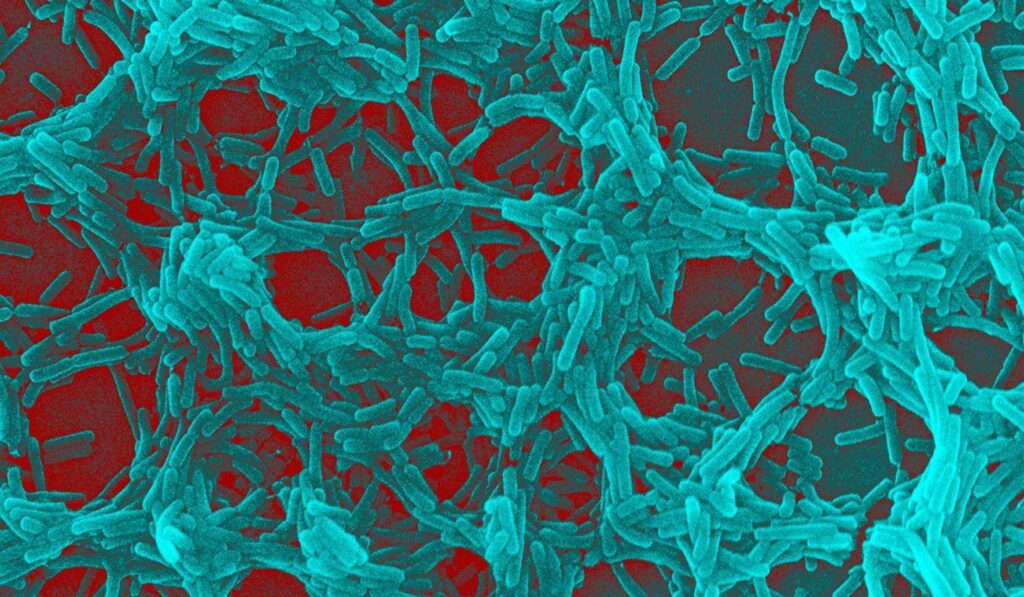
The bacteria living in your gut may hold clues about your risk of developing Parkinson’s disease long before any tremors or movement problems begin, according to a new study that researchers say
Researchers are turning their attention to the trillions of microbes in the gut because they seem to carry patterns that appear well before classic Parkinson’s symptoms show up. Instead of waiting for tremors or slowed movement, scientists are comparing gut bacteria from people who later develop Parkinson’s to those who don’t. The idea is not science fiction: the gut-brain connection is real and could offer an earlier window into risk.
The study examined differences in microbiome composition and found consistent shifts in certain bacterial groups among people who later developed Parkinson’s disease. Those shifts show up during the prodromal phase, when non-motor signs like constipation or changes in smell can already be present. Researchers stress that gut signals are clues, not certainties, and that association is not the same as proof of cause.
One of the appealing aspects of a gut-based signal is that stool sampling is noninvasive and scalable, unlike many neurological tests. That opens the door to screening strategies that could flag higher-risk individuals for follow-up with neurologists. At the same time, we cannot ignore variability between labs, diets, geographies, and the methods used to analyze microbial communities.
Scientists are also exploring mechanisms that could link gut bacteria to brain changes, including inflammation, immune activation, and misfolded proteins. The protein alpha-synuclein, which clumps in Parkinson’s patients’ brains, has been detected in the gut in some studies and may travel along nerves to the central nervous system. Those links remain an active area of investigation and should be tested carefully before people change their behavior based on headlines.
For clinicians, the practical question is whether microbiome profiling could one day join other early indicators like REM sleep behavior disorder or loss of smell to build a risk score. Combining multiple markers could sharpen predictions, but that requires large, diverse datasets and standardized lab methods. Right now, microbiome data are noisy and easily influenced by antibiotics, diet, and other medications.
On the treatment front, the finding rekindles interest in microbiome-targeted strategies as potential preventive tools, from dietary interventions to probiotics or fecal transplants. Those ideas are tempting but premature; interventions must be tested in controlled trials to separate placebo effects from real biological benefit. Any move toward clinical use needs clear evidence of safety, efficacy, and durability.
Public health implications depend on replicability and on whether early detection leads to meaningful action that changes outcomes. If early microbiome signals reliably identify a subset of people at elevated risk, trials could test whether interventions delay or prevent disease onset. Until then, the research offers a promising direction rather than a ready-made screening program.
Patients and families should watch the science but avoid panic or overconfidence in early findings, because translating microbiome research into clinical practice is complex and takes time. Scientists are pushing to standardize sampling, expand longitudinal studies, and integrate clinical signs with microbial signatures. The hope is that combining gut data with established early markers will produce practical tools to guide research and, eventually, care.